Enallylpropymal
From Wikipedia, the free encyclopedia
Jump to: navigation, search
 |
|
| Systematic (IUPAC) name | |
|---|---|
|
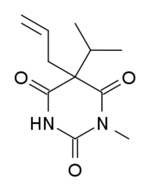
(RS)-1-Methyl-5-allyl-5-isopropyl-1,3-diazinane-2,4,6-trione
|
|
| Identifiers | |
| CAS Number | 1861-21-8 |
| ATC code | none |
| PubChem | CID 95636 |
| ChemSpider | 86329 |
| Synonyms | Enallylpropymal |
| Chemical data | |
| Formula | C11H16N2O3 |
| Molar mass | 224.256 g/mol |
| Chirality | Racemic mixture |
| 3D model (Jmol) | Interactive image |
|
|
|
|
| (verify) | |
Enallylpropymal (Narconumal) is a barbiturate derivative developed by Hoffman la Roche in the 1930s.[1] It has sedative and hypnotic effects and is considered to have a moderate abuse potential.[2]
References[edit]
| Alcohols | |
|---|---|
| Barbiturates |
|
| Benzodiazepines |
|
| Carbamates | |
| Flavonoids |
|
| Imidazoles | |
| Kava constituents | |
| Monoureides | |
| Neuroactive steroids |
|
| Nonbenzodiazepines |
|
| Phenols | |
| Piperidinediones | |
| Pyrazolopyridines | |
| Quinazolinones | |
| Volatiles/gases |
|
| Others/unsorted |
|
|
See also: GABAergics
|
|
| This sedative-related article is a stub. You can help Wikipedia by expanding it. |
Hidden categories: