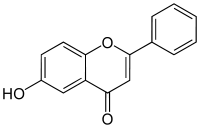
6-Hydroxyflavone
From Wikipedia, the free encyclopedia
 |
|
 |
|
| Names | |
|---|---|
| IUPAC name
6-hydroxy-2-phenylchromen-4-one
|
|
| Other names
6-Monohydroxyflavone; 6-Hydroxy-2-phenyl-4-benzopyrone
|
|
| Identifiers | |
| 6665-83-4 |
|
| ChEMBL | ChEMBL138649 |
| ChemSpider | 65233 |
| EC Number | 229-704-8 |
| Jmol 3D model | Interactive image Interactive image |
| KEGG | C14137 |
| PubChem | 72279 |
|
|
|
|
| Properties | |
| C15H10O3 | |
| Molar mass | 238.24 g·mol−1 |
| Melting point | 234 to 236 °C (453 to 457 °F; 507 to 509 K) |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
|
|
| Infobox references | |
6-Hydroxyflavone is a flavone, a type of chemical compound. It is one of the noncompetitive inhibitors of cytochrome P450 2C9. It is reported in leaves of Barleria prionitis Linn. (a common Acanthaceae from India).[1] 6-Hydroxyflavone may have a potential as a therapeutic drug capable for the treatment of anxiety-like disorders.[2]
References[edit]
- ^ Medicinal plants, Chemistry and properties by M Daniel.
- ^ "GABAA receptor subtype selectivity underlying anxiolytic effect of 6-hydroxyflavone". ScienceDirect. Retrieved 2011-06-12.
External links[edit]
| This article about a phenol is a stub. You can help Wikipedia by expanding it. |