Hexestrol
From Wikipedia, the free encyclopedia
 |
|
| Clinical data | |
|---|---|
| Routes of administration |
Oral |
| Identifiers | |
|
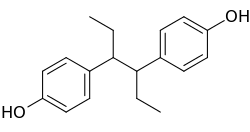
Systematic (IUPAC) name: 4-[4-(4-Hydroxyphenyl)hexan-3-yl]phenol
|
|
| Synonyms | NSC-9894 |
| CAS Number | 84-16-2 |
| PubChem (CID) | 3606 |
| ChemSpider | 3480 |
| Chemical and physical data | |
| Formula | C18H22O2 |
| Molar mass | 270.36608 g/mol |
| 3D model (Jmol) | Interactive image |
|
|
|
|
Hexestrol (INN) (brand name Synestrol, Synoestrol, Estrifar, Estronal, numerous others), also known as hexanestrol, hexoestrol, and dihydrodiethylstilbestrol, is a synthetic, non-steroidal estrogen of the stilbestrol group related to diethylstilbestrol that was used to treat estrogen deficiency but is now no longer employed medically.[1][2][3] Hexestrol has also been available and used in ester form, including as hexestrol diacetate, hexestrol dicaprylate, hexestrol diphosphate, and hexestrol dipropionate.[1] Hexestrol has approximately 302% and 234% of the affinity of estradiol at the ERα and ERβ, respectively.[4]
See also[edit]
References[edit]
- ^ a b J. Elks (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 162–. ISBN 978-1-4757-2085-3.
- ^ I.K. Morton; Judith M. Hall (6 December 2012). Concise Dictionary of Pharmacological Agents: Properties and Synonyms. Springer Science & Business Media. pp. 140–. ISBN 978-94-011-4439-1.
- ^ John A. Thomas (12 March 1997). Endocrine Toxicology, Second Edition. CRC Press. pp. 144–. ISBN 978-1-4398-1048-4.
- ^ Kuiper GG, Carlsson B, Grandien K, Enmark E, Häggblad J, Nilsson S, Gustafsson JA (1997). "Comparison of the ligand binding specificity and transcript tissue distribution of estrogen receptors alpha and beta". Endocrinology. 138 (3): 863–70. doi:10.1210/endo.138.3.4979. PMID 9048584.
| This drug article relating to the genito-urinary system is a stub. You can help Wikipedia by expanding it. |