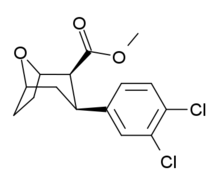
Tropoxane
 |
|
| Clinical data | |
|---|---|
| ATC code | none |
| Identifiers | |
|
|
| PubChem (CID) | 9796921 |
| Chemical and physical data | |
| Formula | C15H16Cl2O3 |
| Molar mass | 315.192 g/mol |
| 3D model (Jmol) | Interactive image |
|
|
| (verify) | |
Tropoxane is an aryloxytropane derivative drug developed by Organix Inc.,[1] which acts as a stimulant and potent dopamine and serotonin reuptake inhibitor. It is an analogue of dichloropane where the amine nitrogen has been replaced by an oxygen ether link (at the bridgehead position), demonstrating that the amine nitrogen is not required for DAT binding and reuptake inhibition.[2][3][4]
P. Meltzer, et al. made some 8-thiabicyclo(3.2.1)octanes recently (2007).
For SAR purposes, a table has been included that lists some of these compounds.
| X | Com | DAT | SERT | Com | DAT | SERT | Com | DAT | SERT |
|---|---|---|---|---|---|---|---|---|---|
| H | 1a | 910 | >10uM | 2a | 140 | >8uM | 3a | 117 | >3uM |
| F | 1b | 220 | >30uM | 2b | 59 | >11uM | 3b | 38 | 494 |
| Cl | 1c | 13 | >10uM | 2c | 11 | 1uM | 3c | 9.6 | 33 |
| Br | 1d | 9.1 | >25uM | 2d | 6.0 | 342 | 3d | 6.0 | 14 |
| I | 1e | 6.7 | >8uM | 2e | 9.0 | 70 | 3e | 14 | 10 |
| Cl2 | 1f | 4.5 | >3uM | 2f | 6.9 | 99 | 3f | 5.7 | 8.0 |
| BN | 1g | 8.0 | >1uM | 2g | 8.0 | 36 | 3g | 16 | 13 |
It had been hypothesized that transporter binding of the tropanes might include ionic bonding of the central tropane nitrogen. But it turned out that at this site neither ionic nor hydrogen bonding is a prerequisite for potent monoamine reuptake inhibition. Oxa- and thia-analogs of RTI-111 are potent inhibitors, and even an N-replacement by methylene holds the potency within the same magnitude.[5][6][7] However, N-quaternisation (N-dimethyl) considerably reduces DAT affinity.
2[edit]
In this SAR, the focus is on seeing the effect of changing 8-NMe to S, O, or CH2.
Both enantiomers, as well as the racemates are presented in several cases for comparison.
| Enant. | X | Com | DAT | SERT | Com | DAT | SERT | Com | DAT | SERT |
|---|---|---|---|---|---|---|---|---|---|---|
| Rac | S | 1a | 4.5 | 3,600 | 2a | 6.9 | 99 | 3a | 5.7 | 8.0 |
| Rac | O | 1a | 10 | 6,000 | 2a | 3.1 | 64.5 | 3a | 3.3 | 6.5 |
| 1R | NMe | 1a | 1.2 | 867 | 2a | 0.4 | 27 | 3a | 1.1 | 2.5 |
| Rac | CH2 | 1a | 7.1 | 5,160 | 2a | 13 | 166 | 3a | 9.6 | 33 |
See also[edit]
References[edit]
- ^ Bertha K Madras and Peter C Meltzer. "Bridge-substituted Tropanes and Uses". WIPO Patent WO/1997/040859
- ^ Madras BK, et al. "Nitrogen-based drugs are not essential for blockade of monoamine transporters". Synapse. 1996 Dec;24(4):340-8. PMID 10638825
- ^ Meltzer PC, et al. "2-Carbomethoxy-3-aryl-8-oxabicyclo[3.2.1]octanes: potent non-nitrogen inhibitors of monoamine transporters". Journal of Medicinal Chemistry. 1997 Aug 15;40(17):2661-73. PMID 9276012
- ^ Madras BK, et al. "Molecular and regional targets of cocaine in primate brain: liberation from prosaic views". Addiction Biology 2006;5(3):351-359.
- ^ Pham-Huu D-P, et al. "Synthesis of 8-thiabicyclo[3.2.1]octanes and Their Binding Affinity for the Dopamine and Serotonin Transporters". Bioorg Med Chem. 2007, 15(2): 1067–82. PMID 17070057 [1]
- ^ Madras BK, et al. "Non-amine-based dopamine transporter (reuptake) inhibitors retain properties of amine-based progenitors". Eur J Pharmacol. 2003; 479(1-3): 41-51. doi:10.1016/j.ejphar.2003.08.055 PMID 14612136
- ^ Compare PMID 11746710 Goulet M, et al. "Non-amines, drugs without an amine nitrogen, potently block serotonin transport: novel antidepressant candidates?" Synapse 2001, 42(3): 129-40.

